New Generation of Cross Link Hyaluronic Acid Filler
Technology
Volomis is a “Made in Italy line” and assumes control over the entire production line and excellent quality. DERMOAROMA GROUP had already ‘shown a strong capacity’ to gain global leadership in niche markets but above all great flexibility ‘in the creation of products closer to the needs of patients and specialists, highly tolerated formulations, practically devoid of side effects (exempt of inflammatory and allergic reactions) and other free toxic substances.
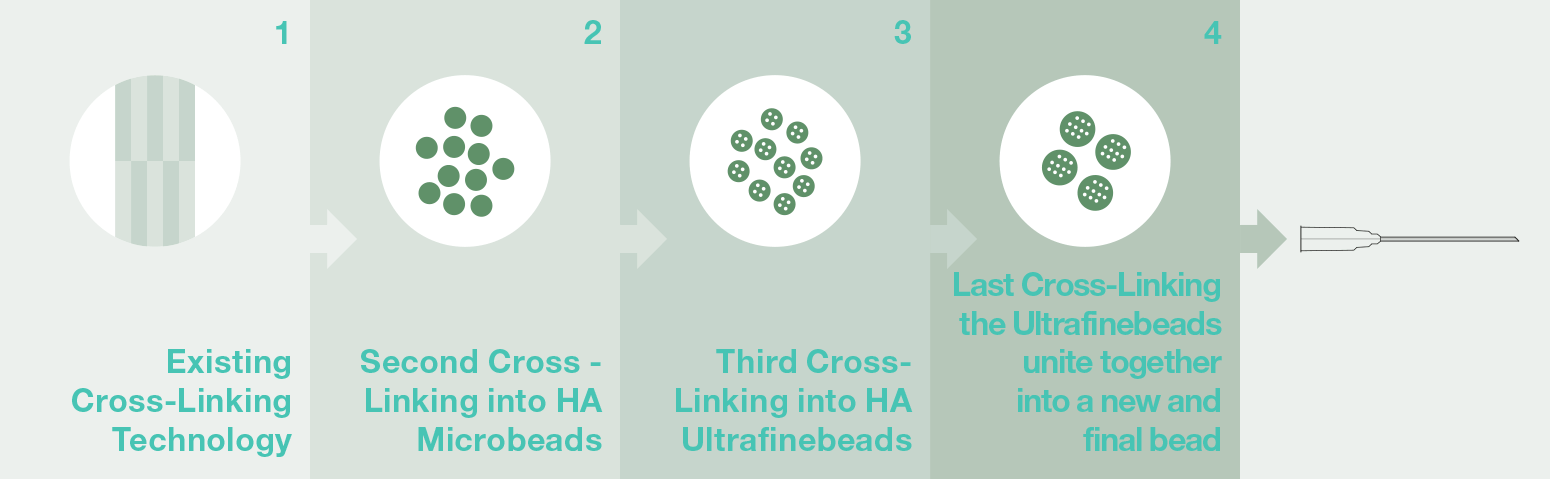
VOLOMIS HA Fillers use a high-quality Hyaluronic acid to create a product that shows an increased Cross-Linking rate. Our product overcomes the conventional fillers on the market due to the special multi-staged cross-linking technology used to create three-layered small beads that resolve the difficulty of creating a long-lasting HA Filler. Volomis is world’s first “long-duration” hyaluronic acid filler that has increased the entry barriers by applying core technology so that the HA filler does not transform its component after product completion and it makes it perfectly safe for the human body.
This manufacturing process does not use raw materials of animal derivation and has eliminated the use of organic solvents at all stages of production materials.
Volomis Features & Characteristics
Long-lastingThanks to the Multi Staged Cross-linking Technology Dermoaroma achieved in creating a Dermal Filler of 4 stage cross-link through the creation of microbeads. This allows for a long-lasting product with high viscosity and cohesiveness and can resist degradation better with time. This characteristic allows not only to obtain an optimal volume effect with less quantity but also slows down the degradation process. This unique characteristic results in avoiding repeated and short time injections (which could be possible risks of complications) are no longer necessary.

Technology in existing HA filler: Primary or Secondary cross-linking can be different in each company.
Volume enhancementVolomis shows exceptional volume enhancement due to its outstanding hydrophilic capacity through its stable particle structure. Moreover, even with a small amount, the product allows for easy molding and with a greater volume enhancement effect. Our product successfully achieved a high viscous semi-solid Hyaluronic acid gel through the Multi-staged Cross-linking Technology overcoming the technological limitations of conventional filler products. It allows for easy molding due to its high viscosity and small particle size.
Safe and effectiveVolomis is a new generation of dermal filler, which sets a higher standard in the market of cross-link hyaluronic acid. Dermoaroma Group, with the technology used to manufacture Volomis family of products based on research and development of the product, is enjoying great success.
The Volomis family of products manufactures under strict quality controls to meet and surpass international standards.

FAQ
The high degree of cross-linking supports a slower degradation of Volomis products in the tissue therefore Volomis products persist at least 6 to 12 months. The durability in the skin varies according to skin type, age and the way of life of the patient as well as treated area and injection technique. It can be necessary to renew the treatment partially in order to optimize the results and durability of the correction.
Volomis products should not be used on pregnant woman or breastfeeding, patients presenting an auto-immune disease, antecedents of allergy or over-sensitiveness to the hyaluronic acid, or an immune-therapeutic treatment also Volomis products should not be injected into inflammatory and/or infectious skin (acne, herpes, impetigo…), a tendency to develop hypertrophic scars.
It should not be used in conjunction with any laser treatment, chemical peeling or dermabrasion. Prior consultation of the patient is necessary to know the medical history, especially aesthetic treatments (like prior injections) and to determine if Volomis products are suitable.
As with all skin fillers swelling and/or redness, with or without associated pain, may occur after the injection. These generally disappear within a few hours, days or in some cases within a week. Some rare cases of necrosis, abscess, granuloma or of over-sensitivity were described in the scientific literature after injections of hyaluronic acid.
Continuation of these reactions or appearance of any other side effects must be reported to the practitioner as soon as possible in order to resolve it by appropriate treatment. The distributor should also be informed about this incident. Before treatment, please inform your patient about the potential side effects related to this injectable implant.
SIGN UP FOR OUR NEWSLETTER
to receive Dermoaroma news & product offers